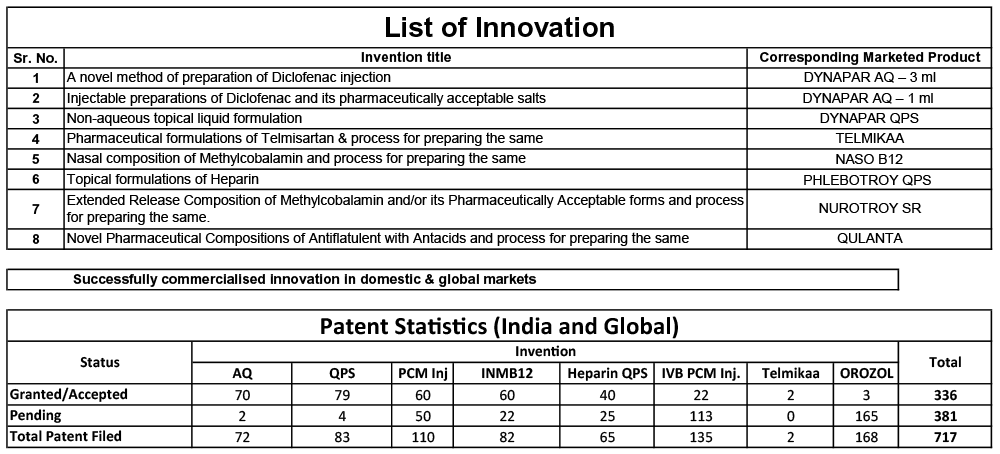
A) Strong portfolio of Patented Innovations:
Our R&D Department is approved by the Department of Scientific & Industrial Research, Government of India and has an innovation-driven approach. This approach has helped us in creating path-breaking brands.
Each patentedbrand at Troikaa has stemmed from original innovations;of which the lead time from concept to commercialization has been effectively maintained from 6 to 10 years. It may affect our short-term profitability but it drives long-term growth.Our R&D innovations have helped steer us into global markets and made us a well-recognized company worldwide.
B) Strong Portfolio of Difficult to Manufacture Products:
Many of Troikaa’s products like Parenteral Emulsions (Propofol; Etomidate injection), inhalation agents for anaesthesia (Isoflurane, Sevoflurane), Sublingual Tablets, etc. have been created after extensive research, dedicated expert manpower, thorough and stringent clinical trials. This has helped create a niche in the industry,which has spearheaded our rapid sales.